WRITE-UP FOR PMR FELLOWSHIP FROM MAY 2022 - PRESENT

Reshma Babu
WRITE-UP FOR PMR FELLOWSHIP FROM MAY 2022 - PRESENT
PhD Topic: 3d- Transition Metal Catalyzed Activation of Alcohols: Applications in Acceptorless Alcohol Dehydrogenation and Related Reactions
Chapter 1
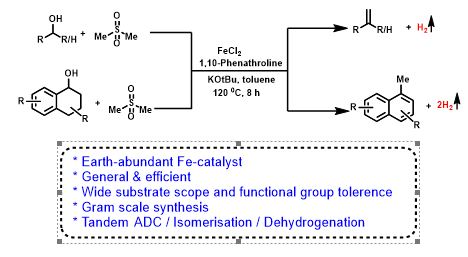
Iron Catalyzed Direct Julia-Type Olefination of Alcohols
Olefins are valuable for synthetic chemistry and ubiquitous in polymer, agrochemical, pharmaceutical, and functional materials. Herein, we have carried out the first iron-catalyzed convenient, and expedient strategy for the synthesis of styrene and naphthalene derivatives with the liberation of dihydrogen. The present olefination has a broad substrate scope with primary and secondary alcohols. Interestingly, unprecedented synthesis of 1-methyl naphthalenes proceeds via tandem methenylation/double dehydrogenation was reported.
Chapter 2:
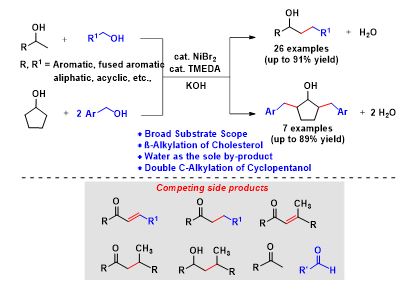
Nickel-Catalyzed Guerbet Type Reaction: C–Alkylation of Secondary Alcohols via Double (de)hydrogenation
The construction of functionalized diverse molecules from simple abundant chemicals by C‒C bond-forming reactions is one of the fundamental reactions in organic synthesis. Herein, we have done the selective β-alkylation of secondary alcohols with primary alcohols, using a commercially available, inexpensive NiBr2/TMEDA as an efficient catalytic system through acceptorless double dehydrogenative cross-coupling strategy. Interestingly, more challenging substrate conversions were achieved under benign conditions using primary alcohols as potential alkylating agents with various secondary alcohols. A broad range of substrates including aromatic, cyclic, acyclic, and aliphatic alcohols was well tolerated. Interestingly, the C-alkylation of cholesterol derivatives and the double C-alkylation of cyclopentanol with various alcohols were also demonstrated.
Chapter 3
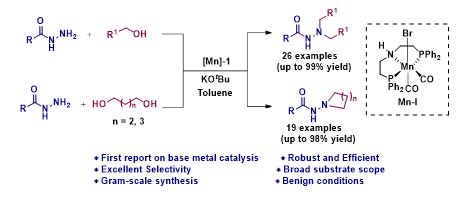
Manganese Catalyzed Double Borrowing Hydrogenation: N,N-Dialkylation and Cyclization of Acylhydrazides Using Alcohols
N, N-disubstituted acyl hydrazide and its derivatives are privileged synthetic scaffolds that have been widely used for the synthesis of agrochemicals, polymers, pharmaceuticals, and bio-active functional materials. Herein, we report the first earth-abundant base metal catalyzed direct one-pot N,N-dialkylation and cyclization of acylhydrazides using alcohols. The reaction is catalyzed by a well defined Mn(1)-based PNP-pincer complex and it operates via the borrowing hydrogen strategy. Surprisingly, more challenging substrates were tolerated under standard reaction condition using alcohols as potential alkylating agents. Interestingly, really demanding cyclization of acylhydrazides with diols also demonstrated.
Chapter 4
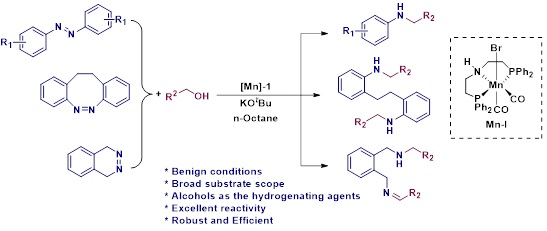
Efficient Manganese Catalyzed Hydrogenation and N-alkylation of Azo (N=N) Bonds to Alkylated Amines Using Alcohols
N-alkylated amine derivatives have wide spread applications in agrochemicals, pharmaceuticals, lubricants, organic dyes, corrosion inhibitors, surfactants and polymer industry. Herein, we systematically explore complete hydrogenation and N-alkylation of azo (N=N) bonds to alkylated amines by using a well-defined Mn-PNP pincer complex via metal-ligand incorporation strategy. Interestingly, the reaction involves abundantly available renewable feedstocks such as alcohols, that can act as hydrogenating as well as the alkylating agents which makes this present work an eco-friendly, atom-economical, step-economical and sustainable approach for alkylated amine synthesis. Moreover, really demanding and more challenging N-alkylation of cyclic diazocines also performed. Also, we have explored the synthesis of N-CH3/N-CD3 amine derivatives.

Chapter 5
Nickel catalyzed synthesis of N-alkylated amine derivatives from phenylhydrazenes with the liberation of ammonia using alcohols as alkylating agent
N-alkylated amine derivatives have wide range of applications in pharmaceutical, agrochemical, dye, resin and electronic material industries. Because of its relevance various methods have been developed to synthesize N-alkylated amine derivatives. Herein, we reported the efficient nickel catalyzed synthesis of N-alkylated amine derivatives from phenylhydrazenes with the liberation of ammonia using alcohols as alkylating agent via reductive N-N bond cleavage strategy. Interestingly, aromatic, fused aromatic, aliphatic, cyclic and acyclic alcohols were well tolerated under standard condition. Also, we have explored one-pot synthesis of symmetrical azines from both primary and secondary alcohols and hydrazene hydrate.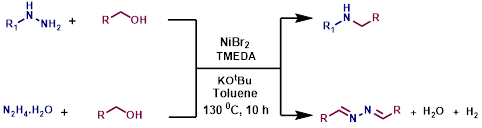
Chapter 6
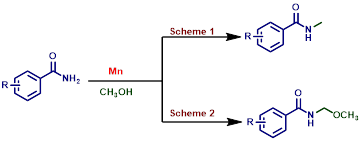
Manganese catalyzed synthesis of N-methylated amides and N-(methoxyalkyl)benzamides
N-(methoxyalkyl)benzamides are very much important because it occurs in insect toxin pederin and in a number of synthetic herbicides. Herein, we have carried out the unprecedented manganese catalyzed convenient, and expedient strategy for the synthesis of N-(methoxyalkyl)benzamide derivatives. The present strategy has a broad substrate scope with methanol as one of the reagents to synthesise N-(methoxyalkyl)benzamides. Interestingly, synthesis of N-methylated amides was also reported just by tuning the standard reaction condition.
External Teaching Assistantships
- Coordination Chemistry and Spectroscopy (At Sri Venkateshwara College of Engineering, TIRUPATI)
Achievements
- Academic Excellence Award
- Delivered a talk on Mole Day symposium, 23rd October 2021 at IISER-Tirupati.
- Qualified PMRF review held on 28th December 2021 with ‘very good’ grade.
- Qualified PMRF review held on 27th June 2022 with ‘Progress satisfactory; continue fellowship’ comment.
Publications
- Rana, R. Babu, M. Subaramanian and E. Balaraman*, Ni-catalyzeddehydrogenative coupling of primary and secondary alcohols with methyl-N-heteroaromatics, Org. Chem. Front., 2018, 5, 3250–3255.
- Landge, R. Babu, V. Yadav, M. Subramanian, Gupta, V, and E. Balaraman*, Iron-Catalyzed Direct Julia-type Olefination of Alcohols, J. Org, Chem., 2020, 85, 9876−9886.
- Babu, M. Subaramanian, S. P. Midya and E. Balaraman*, Nickel-catalyzed Guerbet type reaction: C-alkylation of secondary alcohols via double (de)hydrogenation, Org. Lett., 2021, 23, 3320–3325.
- P. Midya, M. Subaramanian, R. Babu, V. Yadav, and E. Balaraman*, Tandem acceptorless dehydrogenative coupling-decyanation under nickel catalysis, J. Org. Chem. 2021, 86, 7552-7562.
- Reshma Babu, Subarna S Padhy, Ganesan Sivakumar, and Ekambaram Balaraman* Expedient Tandem Dehydrogenative Alkylation and Cyclization Reactions Under Mn(I)Catalysis,Sci.Technol., 2023, DOI; https://doi.org/10.1039/D3CY00009E
Papers in preparation
- Babu, E. Balaraman* Nickel catalyzed synthesis of N-alkylated amine derivatives from phenylhydrazenes with the liberation of ammonia using alcohols as alkylating agent (Manuscript under preparation)
- R. Babu and E. Balaraman*, Manganese catalyzed synthesis of N-methylated amides and N-(methoxyalkyl)benzamides (Manuscript under preparation)


